HPC User Report from J. Martens (Comprehensive Heart Failure Center / Universitätsklinikum Würzburg)
Computational Cardiology
Computational Fluid Dynamics simulations of blood flow and contrast agent transport are performed on authentic cardiovascular 3D-geometries. Aim is the analysis of systematic errors in quantitative perfusion measurements [1-4] with the help of contrast-enhanced magnetic resonance imaging.
Motivation and problem definition
In dynamic contrast-enhanced MRI perfusion measurements, passage of intravenously injected contrast agent (CA) through tissue is monitored to quantify myocardial blood flow (MBF). This requires knowledge of the shape of CA wash-in through upstream coronary vessels, the arterial input function (AIF). For technical reasons this is measured in the left ventricle, which introduces risks of systematic errors in MBF quantification due to bolus dispersion in coronary vessels. To analyze and quantify this effect Computational Fluid Dynamics (CFD) simulations of blood flow and CA transport are performed. To guarantee physiological relevance boundary conditions must be defined carefully [5].
Methods and codes
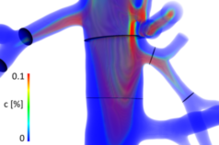 With dedicated software (SimVascular, www.simvascular.org) 3D-models of coronary arteries are extracted from medical imaging data. After discretization with a computational grid (cfMesh, creativeFields), consecutive simulations of blood flow and CA transport are performed with CFD software OpenFOAM (www.openfoam.org). Analogous to electrical circuits, an alternate boundary condition (BC), which comprehends tissue as an electrical capacitor [6], integrates myocardial pressure acting on cardiac vessels into the simulations. The computation jobs are performed on LiMa parallelized on 24 processors. Simulations of both blood flow and CA transport take ~4 days altogether.
With dedicated software (SimVascular, www.simvascular.org) 3D-models of coronary arteries are extracted from medical imaging data. After discretization with a computational grid (cfMesh, creativeFields), consecutive simulations of blood flow and CA transport are performed with CFD software OpenFOAM (www.openfoam.org). Analogous to electrical circuits, an alternate boundary condition (BC), which comprehends tissue as an electrical capacitor [6], integrates myocardial pressure acting on cardiac vessels into the simulations. The computation jobs are performed on LiMa parallelized on 24 processors. Simulations of both blood flow and CA transport take ~4 days altogether.
Results
We have devised and optimized an alternate BC, which yields qualitatively improved blood flow behavior with increased blood flow in diastole compared to systole compared to previously used BC [5]. It allows CFD simulations of blood flow with application of realistic pressure time-curves at the model inlet. Subsequent CA transport simulations show MBF underestimation between 7-21% due to CA bolus dispersion within the model. These results are in agreement with previous findings [1-5]. However, the range of the results as well as their sensitivity to the choice of BC parameters show that fine-tuning is still necessary. Nonetheless, a detailed analysis of particle transport in general (medication, CA, vasodilators etc.) within coronary vessels to different regions of interest remains of high interest.
Outreach
The obtained results will be part of a manuscript in preparation. The performed analyses are part of the BMBF grants 01EO1004, 01E1O1504 and the optimization of the new BC may be part of a following DFG-proposal.
References
- Graafen D, Hamer J, Weber S, Schreiber LM: Quantitative myocardial perfusion magnetic resonance imaging: impact of pulsatile flow on contrast agent bolus dispersion. Physics in Medicine and Biology, 56 (2011) 5167-5185.
- Sommer K, Schmidt R, Graafen D, Breit HC, Schreiber LM: Resting myocardial blood flow quantification using contrast-enhanced magnetic resonance imaging in the presence of stenosis: A computational fluid dynamics study, Medical Physics, 42 (7), 4375-4384, 2015.
- Martens J, Panzer S, van den Wijngaard JPHM, Siebes M, Schreiber LM. Analysis of Coronary Contrast Agent Transport in Bolus-Based Quantitative Myocardial Perfusion MRI Measurements with Computational Fluid Dynamics Simulations. 9th international conference on Functional Imaging and Modeling of the Heart (FIMH 2017), Toronto, Canada; 11.-13.6.2017, Springer LNCS 10263 (369-380).
- Calamante F, Willats L, Gadian DG, Connelly A. Bolus Delay and Dispersion in Perfusion MRI:
Implications for Tissue Predictor Models in Stroke. Magnetic Resonance in Medicine. 2006. 55, 1180-1185. - Sommer K, Bernat D, Schmidt R, Breit HC, Schreiber LM: Contrast Agent Bolus Dispersion in a Realistic Coronary Artery Geometry: Influence of Outlet Boundary Conditions, Annals of Biomedical Engineering, 42 (4), 787-796, 2013.
- Kim HJ, Vignon-Clementel IE, Coogan JS, Figueroa CA, Jansen KE, Taylor CA: Patient-Specific Modeling of Blood Flow and Pressure in Human Coronary Arteries. Annals of Biomedical Engineering, 38 (10), 3195-3209, 2010.
Researcher’s Bio and Affiliation
Johannes Martens received his “Diploma” in Physics in Bonn, Germany (2011). After 3 years as a technical assessor at the “Gesellschaft für Anlagen und Reaktorsicherheit” (GRS) in Cologne, Germany, he started work at the Comprehensive Heart Failure Center (CHFC) in Würzburg, Germany, in 2015 as a PhD student at Prof. Laura Schreiber’s Chair for Cellular and Molecular Imaging.
